Malaria
Global Impact
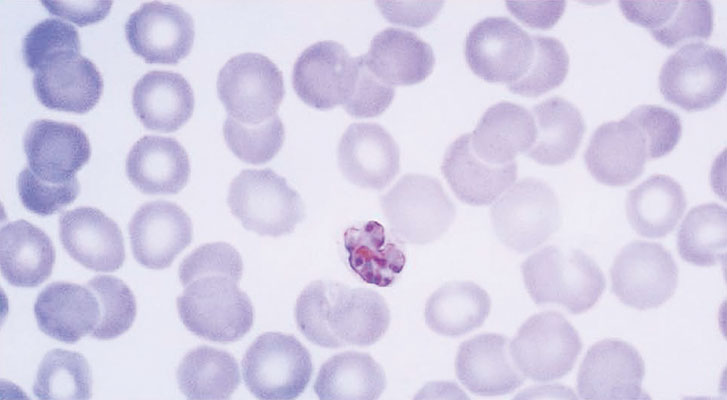
One of humanity’s most historically significant diseases, and still today a major cause of worldwide mortality, malaria results from the transmission of the single-cell parasite Plasmodium via the bite of an infected Anopheles mosquito. Malaria is primarily a disease of the tropical and subtropical regions of the world. About 3.2 billion people – almost half of the world’s population – are at risk of malaria and more than 200 million people are affected by malaria each year, resulting in an estimated 584,000 malaria deaths. At least 90% of those deaths were in Africa, with the heaviest mortality concentrated in children under the age of 5.
Impact in Southeast Asia
Southeast Asia by comparison has seen considerable reductions in malaria cases over the course of the past decades. Nevertheless, malaria still has a serious impact on this region. The WHO reports that in 2010 there were 4.3 million cases and 2,426 deaths due to malaria in Southeast Asia. Most malaria infections are caused by either Plasmodium falciparum or P. vivax and are concentrated among those living on the border of Thailand and in neighboring countries where the disease most affects poor and vulnerable populations, such as migrant workers, subsistence farmers, and minority communities.
World Leader
FTM has been leading research into the disease in Asia for over 50 years, and is currently engaged in many projects and collaborations to bring about the elimination of malaria. The Faculty has provided a significant proportion of the biological, economic and clinical basis for the change in global antimalarial treatment recommendations to artemisinin combination treatments. We have been responsible for publishing 9.4% of all antimalarial trials since 1966, and enrolled 22% of all patients in such studies worldwide. In the period 2009-2013, Mahidol University was ranked 5th in the world with regards to publications on malaria research.

Largest Trial
In 2005 the South East Asian Quinine Artesunate Malaria Trial (SEAQUAMAT) group, which included members from MORU (Mahidol Oxford Tropical Medicine Research Unit) published results the largest ever trial in severe malaria. The multi-country prospective study with 1,461 patients compared mortality in patients treated with intravenous artesunate or intravenous quinine. The trials provided definite proof artesunate should become the treatment of choice for severe falciparum malaria in adults. This recommendation was endorsed by the World Health Organization (WHO) and included in its malaria treatment guidelines.
Characterization of Artemisinin Resistance
One major challenge in malaria treatment and elimination is the growth of artemisinin resistance, a worrying trend that has been extensively mapped by the Mahidol-Oxford Tropical Medicine Research Unit’s (MORU) TRAC project (Tracking Resistance to Artemisinin Collaboration) which involved 15 sites across 10 countries. Artemisinin based drugs have been key components in the treatment of malaria since the antimalarial properties of the substance was first discovered here at FTM, and emerging resistant strains increases the demand for new drug development and methods of stopping the spread of these resistant strains. The TRAC project was the first comprehensive phenotypic and genotypic characterization of artemisinin resistance in falciparum malaria.
Work on malaria at the Faculty be grouped into the following main categories:
Epidemiology and data management
In Thailand, malaria has been reduced significantly, and is currently only endemic in the border regions, primarily on the Thai-Myanmar border. Treatment in these areas is of high priority, and yet remains difficult due to a large proportion of migrant workers and refugees living in inaccessible areas where the public health system struggles to reach. The Faculty has been involved in these areas for a long time, based out of the Tropical Diseases Research Center (TDRC) in Kanchanaburi Province, and the Rajanagarindra Tropical Disease International Center (RTIC) in Ratchaburi Province. These centres are run by FTM and serve as labs for the Faculty’s researchers as well as clinics that provide healthcare services for locals.
Diagnostics
Fast, reliable, and cheap diagnostic methods are crucial both in order to provide reliable epidemiological information, and to ensure early treatment against malaria, which increases speed and likelihood of recovery. Diagnosis can also tell us which type of malaria infection patients have contracted, and what genetic composition the parasites have. This information allows us to develop drugs that target common genetic components, and reveals correlations where certain types of patients tend to contract specific types of infections. Researchers at the Faculty have been studying the molecular and genetic components of the different strains of malaria and strive to develop ever more effective diagnostic methods.
Vector control
Malaria is spread by the Anopheles mosquito, and understanding its life-cycle increases our ability to effectively control malaria spread. Researchers at the Faculty’s Department of Entomology have been tracking and monitoring mosquito activities in the border regions of Thailand-Myanmar and Thailand-Cambodia, cataloging and mapping mosquito behavior. One part of this mapping effort is focused on tracking different mosquito species, as different species are susceptible to different malaria parasites, and sensitive to different insecticides. Understanding the relationship between mosquito and parasite can help us develop drugs or insecticides that prevent infection spread. Another focus is to look at mosquito targets, as this also affects the spread of the disease. If we know that certain types of mosquitos favor cattle, humans, or other targets, we are able to develop more effective measures in controlling their ability to spread the disease. Therefore, ongoing monitoring efforts are conducted by the Faculty’s entomologists to monitor mosquito species distribution.
Treatment/Drug development
One of the biggest challenges in the treatment of malaria is the emergence of artemisinin resistant malaria strains, which have been observed in more and more places around the Southeast Asian region. Artemisinin has been used as a key component in malaria treatment. However, recent reports of growing resistance to this treatment create the need for new approaches. Researchers at the faculty strive to find genetic targets that can be used in drug development, testing, and applying treatments in a clinical setting.
Dengue
Significant Mosquito-borne Virus
Dengue virus (DENV) is the single-most significant disease-causing mosquito-borne virus in tropical and subtropical regions of the world today, producing outcomes as varied as mild fever to hemorrhage and shock leading to death. In spite of considerable research, no antiviral drugs are currently available and the only available therapy is essentially supportive. Transmitted by mosquitoes of the Aedes genus, DENV is a member of the Flavivirus genus, a group that includes viruses responsible for Yellow Fever, Japanese Encephalitis, and Zika virus infections.
Increasing Threat
The US CDC estimates that almost 400 million infections occur annually worldwide. Thailand alone recorded some
140,000 cases of infection in 2015, with 126 deaths, and Thailand’s Public Health Ministry suggests that dengue cases may increase further in following years. The causes for the increase in dengue cases are many but certainly include rapid urbanization and more frequent air travel, both of which increase the chances of transmission, the expansion of mosquito breeding habitats, and possibly climate change, which is allowing the mosquito vector to extend its range into new territory.
Vaccine Development
Thai researchers and clinicians have long been in the forefront of efforts to better understand and treat dengue infections and the Faculty of Tropical Medicine has played a particularly key role over the years, perhaps most notably in what is the single most important development in the field of dengue research in the last decade: 2015 saw the commercial licensure of the world’s first dengue vaccine, Dengvaxia (CY D-TDV), a live recombinant tetravalent dengue vaccine developed by the company Sanofi Pasteur. A safe, effective and affordable vaccine against dengue has been long-sought and progress in bringing it about has been fraught with difficulty, but fortunately the goal has been achieved and the Faculty of Tropical Medicine played an important role in making it happen. Researchers from the Department of Clinical Tropical Medicine, the Department of Tropical Pediatrics and the Vaccine Trial Center, together played a key role in testing the vaccine in Thai children between the ages of 2 and 16 years, conducting trials that found the dengue vaccine to be safe, moderately efficacious, with an overall 81% reduction in risk of severe dengue infection. The Dengvaxia vaccine is now on the market for use in children over 9 years old in Mexico, Brazil, and the Philippines, and should become available in Thailand sometime in 2016. Other vaccines against dengue are in the pipeline and work with international collaborators is ongoing at the Faculty of Tropical Medicine in order to improve vaccine efficacy. The BIKEN Endowed Department of Dengue Vaccine Development is focusing on chimeric DNA vaccines, which have the advantage of remaining stable at warm temperatures, have lower manufacturing costs, and offer greater security, because live attenuated viruses are more likely to change and regain their virulence.

Researching Treatment
Developing effective drug treatments for dengue infection is challenging. Most individuals are generally asymptomatic during the early stages of infection and often by the time symptoms do appear the body’s immune system has already begun clearing the virus from the system. This then presents a very narrow window in which treatment can be of any utility. Fortunately, increasingly accurate diagnosis allows clinicians to identify those patients most at risk for serious disease. Nevertheless, a safe and effective treatment for dengue infection would be a tremendous boon to physicians and patients alike. Researchers with the Department of Social and Environmental Medicine have collaborated with the Center of Excellence for Antibody Research (CEAR) are investigating the use of monoclonal antibodies, which in addition to their application to vaccines, can be used through passive immunization to protect against dengue infection or reduce virus levels. CEAR researchers demonstrated that mutations introduced into anti-dengue antibodies could both boost their protective effects and reduce possible harmful effects.
Melioidosis
Neglected Tropical Disease
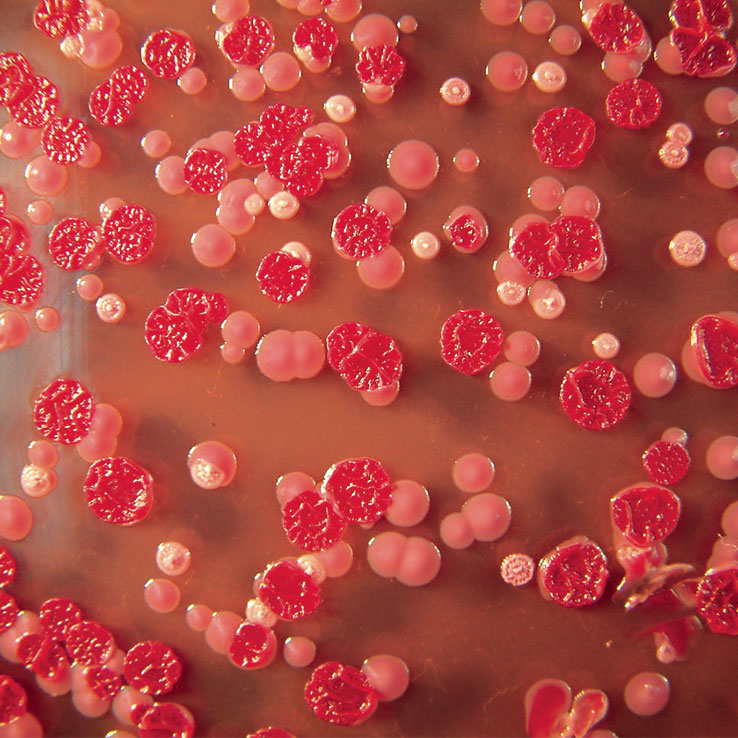
Melioidosis is an infectious disease caused by the flagellated gram-negative bacterium Burkholderia pseudomallei, an environmental saprophyte that is found in soil and water. Infection usually takes place with the inoculation of the bacteria into the body via cuts and wounds in the skin or via inhalation of dust or droplets and very rarely by ingestion of contaminated water. The disease exists in both acute and chronic forms, however, the disease symptoms may vary tremendously, in terms of incubation period, length of illness, and organs involved. Common signs and symptoms can include fever, pain in the chest, bones, or joints; cough; skin infections, lung nodules and pneumonia. A classic “neglected tropical disease”, melioidosis has long been seen as an endemic disease limited to Northern Australia and Southeast Asia but most particularly to northeast Thailand, which has the highest incidence of melioidosis recorded in the world (21.3 cases of melioidosis per 100,000 people per year) and where 80% of children in this region have tested positive for antibodies against B. pseudomallei by the age of four.
Groundbreaking Work
The Faculty of Tropical Medicine has been in the forefront in investigating this disease and it is largely because of this work that we are now beginning to understand that the true global burden of melioidosis is actually greatly underestimated due to the lack of adequate diagnostic microbiological facilities. A groundbreaking paper published in 2015 in the journal Nature Microbiology by Dr Direk Limmathurotsakul and colleagues reports on their review of documented human and animal cases and the records of environmental B. pseudomallei. After combining this data into a formal modeling framework, they estimated the global burden of melioidosis to be severely underreported in the 45 countries in which it is known to be endemic, and predict that the bacteria is probably endemic in a further 34 countries that have never previously reported the disease. The paper, entitled “Predicted global distribution of Burkholderia pseudomallei and burden of melioidosis” has already been widely cited as it is now clear that melioidosis is indeed a major public health threat worldwide.
Cross Department Collaboration
MORU has been working in Sappasithiprasong Hospital in Ubon Ratchathani since 1986. This work has included treating over 3000 patients with melioidosis, as well as research that has helped shape the WHO’s guidelines for treatment of the disease. While the Department of Tropical Hygiene published a study in 2013 on the economic cost of melioidosis in two provinces in Northeast Thailand, revealing the large impact on the communities affected. A collaboration between several different FTM departments has identified a pathogen recognition receptor involved with pathogenesis, which has potential implications in treatments for the disease. The Microbiology and Immunology Department is also working on the disease, and has developed a potential method for rapid detection of the disease in the field.
Helminthiases
Helminths pose significant public-health challenges in many countries of Southeast Asia, with about one-third of the world’s cases of helminth-associated disease occurring in Southeast Asian countries. Although improvements in water and sanitation, along with large deworming campaigns, have sharply reduced prevalence, rural and poorer areas remain at risk.
While the burden of helminthic infections has decreased, there is still much work to be done, particularly in rural and less economically-advantaged parts of Southeast Asia. The Faculty of Tropical Medicine is in the forefront of these efforts, working in many areas to improve the current situation through improvements in diagnosis and the better delivery of treatment.
The Department of Helminthology is the lead department in the Faculty in investigating helminthic infections and is carrying out research across a number of fields, including diagnosis, epidemiology, and taxonomy of both parasite and host. The Department is one of just a few reference labs globally that is able provide a diagnosis for gnathostomiasis and processes samples all over the world. The Department of Tropical Pediatrics, the Department of Protozoology and the Department of Clinical Tropical Medicine are also involved in helminthiases studies.
HIV / AIDS
Global and National Impact
According to 2014 estimates by WHO and UNAIDS, 36.9 million people were living with HIV globally. In the same year, 2 million people became newly infected, and 1.2 million died of HIV-related causes. Although the disease is not confined to tropical regions, many countries in the Tropics are severely affected. After sub-Saharan Africa, the Asia and Pacific region has the largest number of people living with HIV. The World Health Organization estimates that 500,000 people in Thailand are HIV positive. While ARV treatment is effective in some cases with early intervention, a vaccine remains a major goal in the field.
The RV 144 trial
The RV 144 trial, which showed an estimated vaccine efficacy of 31% for protecting low-risk Thai volunteers against the acquisition of HIV-1, was the first ever trial to demonstrate any efficacy of a vaccine regimen. The trial carried out vaccinations in Thailand over the course of 24 weeks starting in October 2003 then tested for HIV until July 2006. The trial was published in the New England Journal of Medicine in 2009.
Centers of Excellence
Two faculty Centers of Excellence are involved with HIV/AIDS research. The Vaccine Trial Center (VTC) is involved in trials of several potential HIV candidate vaccines. In 2012, the VTC began an ongoing stage III trial with an ALVAC-HIV rgp 120 B/E boost vaccine. Researchers at the VTC are also studying the immune response of the host locally at the viral entry site. It is hoped that the findings from this study can further modify the next iteration of the vaccine regimen.
The Center of Excellence for Biomedical and Public Health Informatics (BIOPHICS) has also been conducting studies into HIV, looking at the social issues surrounding vaccine trials. This work is crucial to better understanding factors associated with subjects’ participation in trials, and is useful in terms of reliability and validity of other studies. The center has also been working on Immunology and antibody responses in HIV vaccine trials.
Silom Community Clinic @TropMed
Silom Community Clinic (SCC) moved to the Faculty in 2013. From the outset, the Silom Community Clinic was created as an LGBTQI-friendly sexual health clinic for men who have sex with men (MSM) and transgender women (TGW). Silom Community Clinic integrates research with the provision of services and has been involved in many important research studies, including the 5-year Bangkok MSM Cohort Study and a recent clinical trial assessing the adherence, coverage and behaviors associated with non-daily pre-exposure prophylaxis (PrEP) to prevent HIV infection (HPTN 067).
Neglected Tropical Diseases
Neglected tropical diseases (NTDs) affect over a billion people worldwide, mostly in impoverished populations in developing countries. Global health policies are starting to focus more and more on NTD prevention and control strategies. The Faculty of Tropical Medicine has a number of experts on various NTDs, including the following:
Toxoplasmosis
Toxoplasmosis is a disease caused by the parasitic protozoan Toxoplasma gondii. Although felines are the definitive host, the parasite can infect most warm-blooded organisms, including humans. The protozoan is found worldwide and currently infects up to a third of the global human population, though it is more common in tropical areas. While often asymptomatic, it can be transmitted vertically to cause congenital defects, and can be fatal in immunocompromised patients.
A 2009 study including staff from Siriraj Hospital attributed 8% of AIDS deaths in a cohort to Toxoplasmosis infections. In addition to this, a paper was also released by the Department of Clinical Tropical Medicine, identifying inaccuracies in diagnostic tests for Toxoplasmosis. The combination of a high mortality rate in some groups, congenital defects and little background knowledge of the disease makes FTM’s continued research into Toxoplasmosis very important.
Chikungunya
Chikungunya is a viral disease that causes fever and severe joint pain. It is transmitted to humans by infected mosquitoes. The disease shares similar symptoms with dengue, and in areas where dengue is common misdiagnosis is common. There is no cure for the disease; treatment is focused on relieving the symptoms. The disease occurs in Africa, Asia and the Indian subcontinent and in recent years mosquito vectors of chikungunya have spread to Europe and the Americas. In 2007, disease transmission was reported for the first time in a localized outbreak in north-eastern Italy.
MOCID (Mahidol-Osaka Center for Infectious Diseases) has developed a new rapid diagnostic test kit for chikungunya will finally allow physicians to differentiate this disease from dengue properly, and thus offer more accurate and timely treatment. MOCID have also discovery of possible chikungunya “immune escape variants”, which implies that the ability of the chikungunya virus to evade the human immune system is greater than previously expected.
Scrub typhus
Scrub typhus is an acute, febrile, infectious illness that is caused by Orientia tsutsugamushi. It is endemic to East and Southeast Asia and Northern Australia. The illness often presents as fever and can be difficult to distinguish it from other endemic diseases such as typhoid, leptospirosis, and dengue.
Different departments and units at the Faculty work on Scrub typhus. For example an article published in The Lancet Global Health, in 2015, described work by members of MORU (Mahidol-Oxford Tropical Medicine Research Unit) and international colleagues to find out if scrub typhus, murine typhus and leptospirosis contribute to CNS infections in Lao PDR. The findings established that, in Lao PDR, these neglected diseases are important causes of CNS infections and that antibiotics, such as tetracycline, needed for the treatment of murine typhus and scrub typhus, are not routinely advised for treatment of CNS infections.